FX-rates reduce Australian clinical trial costs
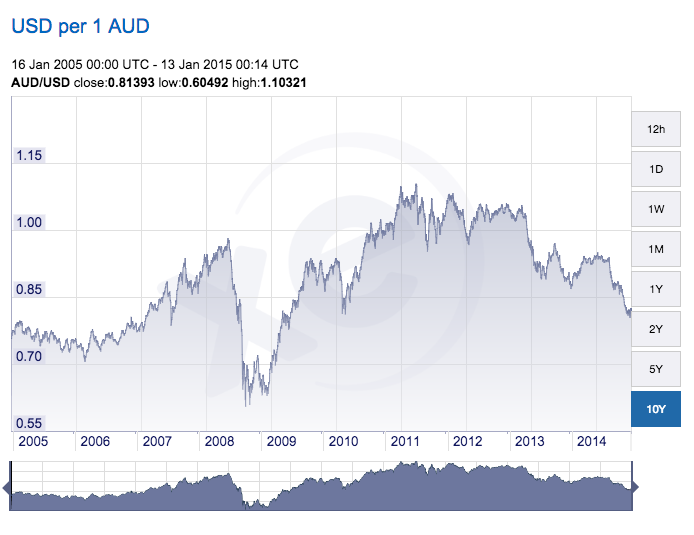
The cost of having clinical trial work done in Australia has moved down dramatically recently and likely to stay down while the China demand for mined raw materials remains low and the Australian economy adjusts to the changes……. USD ~$0.80 buys AUD $1.00.
http://www.xe.com/
The influence of lower oil prices and the prospect of increasing interest rates in the USA while Australian interest rates remain steady or are cut, support the expectation that the Australian Central bank’s wish to see the AUS $ down below the current position, will be realised.
There are significant economic indicators that support a good case for USA clinical research groups to extend their projects to Australia. This may include several elements of the clinical trial process such as data management, analysis and medical writing besides the establishment of sites and the monitoring/quality of patient data progress.
The Datapharm Australia team has a reputation of speedy project start-ups and high quality work. Datapharm’s experience in application of latest technology and sophisticated management of all clinical trial issues, enhances study patient uptake and quality completion on time and budget.
The Australian Government supports the maintenance and development of those elements within the economy that support the transition from the China led mining boom to the services sectors. This suggests that the R&D tax refund system currently in place will remain, although some changes may be made to ensure the objectives of the scheme are still met.
The Australian regulations applied by the TGA remain & support very fast start-up of clinical trials through its notification process (CTN). This means that a Phase I, II, III or IV trial can start within a week, once the ethics committee approval is provided. There are several local ethics committees both private and government and some cover multiple investigator sites. The expertise at Australian clinical trial sites, particularly for Phase 1, is well developed.
With Australia close to Asia time zones allow a greater daily window for communications for running projects more effectively. This means that in essence the access to trial patients for Australia can be expanded easily to the North. Also the population in Australia is a reflection of the world population because it is derived from almost everywhere on the globe.
Best wishes for the new Year!

